|
Putting these pieces together gives the name carbon tetrachloride for this compound. The second element, chlor ine, becomes chlor ide, and we attach the correct numerical prefix (“tetra-”) to indicate that the molecule contains four chlorine atoms. The name begins with the name of the first element-carbon. To write the formula for Carbon tetrachloride we’ll use the Periodic Table and. Let us practice by naming the compound whose molecular formula is CCl 4. In this video well write the correct formula for Carbon tetrachloride (CCl4). *This prefix is not used for the first element’s name. Nomenclature, a collection of rules for naming things, is important in science and in many other situations.This module describes an approach that is used to name simple ionic and molecular compounds, such as NaCl, CaCO 3, and N 2 O 4. 
\): Numerical Prefixes for Naming Binary Covalent Compounds Number of Atoms in Compound 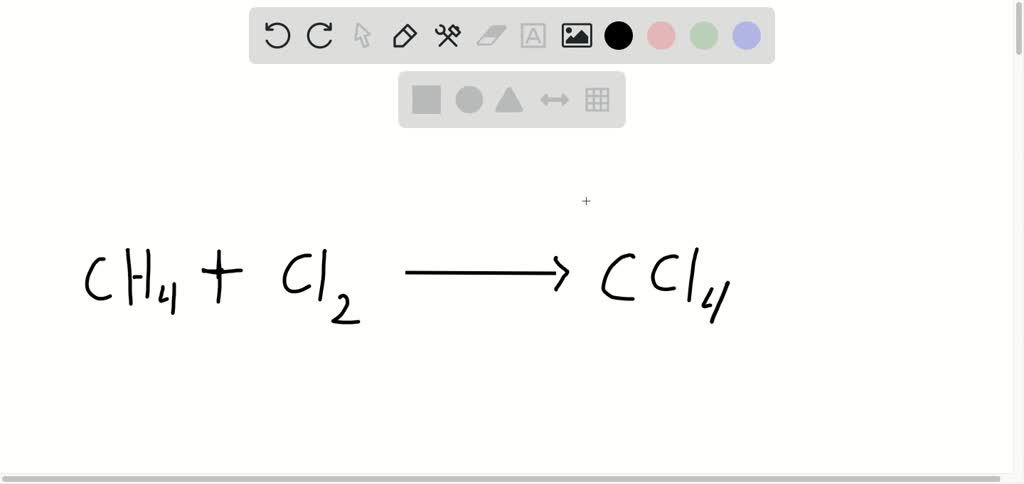
By 2009, only 26 manufacturers worldwide produced carbon tetrachloride, including three in the United States. production and export of CCl 4 dropped precipitously during this time. By 2009, the United States no longer regularly imported CCl 4 only 90 pounds (41 kilograms) were imported during 1996-2009. Question: Problems: Identify the following as Ionic compounds, Covalent compounds, or Acids and then write their chemical formulas: 1. Chemical structure: This structure is also available as a 2d Mol fileor as a computed3d SD file. IUPAC Standard InChIKey:VZGDMQKNWNREIO-UHFFFAOYSA-NCopy. īoth the internationally ratified Montreal Protocol (which first went into effect in 1989) and the United States 1990 Clean Air Act Amendments were instrumental in reducing environmental concentrations of CCl 4 and other ozone-depleting chemicals. IUPAC Standard InChI:InChI1S/CCl4/c2-1(3,4)5Copy. It is a colorless, flammable, neurotoxic liquid that is used as a building block in organic synthesis. It is not permitted in products intended for home use however, chemicals containing CCl 4 can still be purchased online. Carbon disulfide (also spelled as carbon disulphide) is an inorganic compound with the chemical formula CS2 and structure SCS. 2 Write the chemical formula for the following binary compounds given their constituent ions: (a) iron(II) sulfide, Fe. In the United States, CCl 4 use is restricted to industrial and laboratory applications only. Write the chemical formula for the following binary compounds given their constituent ions: (a) copper(I) oxide, Cu + and O. 
Environmental Protection Agency cancelled CCl 4‘s use as a fumigant in 1986. Study with Quizlet and memorize flashcards containing terms like Carbon tetrachloride (CCl4 ) is used as a propellant in aerosol cans. It is a non-flammable, colourless liquid with a 'sweet' chloroform-like smell that can be detected at low levels. CCl 4 was also used as an antihelminthic, a grain fumigant, and a component in fire extinguishers.Ĭarbon tetrachloride’s toxicity was recognized in the early 1900s, and most of the uses were discontinued by the mid-1960s. Carbon tetrachloride, also known by many other names (such as carbon tet for short and tetrachloromethane, also recognised by the IUPAC) is a chemical compound with the chemical formula CCl 4. It was found in household cleaning supplies and spot removers for carpets, clothing, and furniture. In the United States, CCl 4 was also an ingredient in many industrial fluids, and was an effective metal degreaser. Historically, CCl 4 was mainly used to produce chlorofluorocarbons (CFCs), which are used as heat transfer agents in refrigerating equipment and as aerosol propellants.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
